A) dissolving a small amount of solid sodium acetate
B) adding a small amount of dilute hydrochloric acid
C) adding a small amount of dilute sodium hydroxide
D) dissolving a small amount of solid sodium chloride
E) diluting the buffer solution with water
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The solubility of aluminum hydroxide in water ______________ when dilute nitric acid is added to it.
A) increases
B) decreases
C) does not change
D) first increases, then decreases
E) first decreases, then increases
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the [H3O+] in a buffer that consists of 0.30 M HCOOH and 0.20 M HCOONa? For HCOOH, Ka = 1.7 * 10¯4
A) 1.1 * 10¯4 M
B) 2.6 *10¯4 M
C) 4.3* 10¯4 M
D) 6.7 *10¯5 M
E) None of these choices is correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What will be the effect of adding 0.5 mL of 0.1 M NaOH to 100 mL of an acetate buffer in which [CH3COOH] = [CH3COO¯] = 0.5 M?
A) The pH will increase slightly.
B) The pH will increase significantly.
C) The pH will decrease slightly.
D) The pH will decrease significantly.
E) Since it is a buffer solution, the pH will not be affected.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What volume of 0.500 M H2SO4 is needed to react completely with 20.0 mL of 0.400 M LiOH?
A) 4.00 mL
B) 8.00 mL
C) 12.5 mL
D) 16.0 mL
E) 32.0 mL
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the solubility of magnesium sulfate, MgSO4, when placed into a 0.10 M MgCl2 solution. Ksp = 5.9 *10¯3
A) 4.2 * 10¯2 M
B) 5.9 *10¯2 M
C) 7.7 *10¯2 M
D) 3.5 * 10¯5 M
E) 3.5 * 10¯6 M
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The concentration of the complex ion in each of following solutions is 1.00 M. In which of the solutions will the concentration of the uncomplexed metal ion be the greatest? 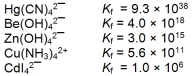
A) Hg2+
B) Be2+
C) Zn2+
D) Cu2+
E) Cd2+
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A buffer is prepared by adding 100 mL of 0.50 M sodium hydroxide to 100 mL of 0.75 M propanoic acid. Is this a buffer solution, and if so, what is its pH?
A) It is a buffer, pH > pKa of propanoic acid.
B) It is a buffer, pH < pKa of propanoic acid.
C) It is a buffer, pH = pKa of propanoic acid.
D) It is a buffer, pH = pKb of sodium propanoate.
E) Since sodium hydroxide is a strong base, this is not a buffer.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pH of a buffer that consists of 0.20 M NaH2PO4 and 0.40 M Na2HPO4? For NaH2PO4, Ka = 6.2*10¯8
A) 6.51
B) 6.91
C) 7.51
D) 7.90
E) 8.13
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A solution is prepared by mixing 50.0 mL of 0.50 M Cu(NO3) 2 with 50.0 mL of 0.50 M Co(NO3) 2. Sodium hydroxide is added to the mixture. Which hydroxide precipitates first and what concentration of hydroxide ions present in solution will accomplish the separation? Ksp = 2.2 *10¯20 for Cu(OH) 2, Ksp = 1.3 *10¯15 for Co(OH) 2
A) Co(OH) 2; [OH¯] = 6.9 * 10¯6 M
B) Co(OH) 2; [OH¯] = 2.6 * 10¯7 M
C) Cu(OH) 2; [OH¯] = 1.8 * 10¯7 M
D) Cu(OH) 2; [OH¯] = 1.1 * 10¯9 M
E) Cu(OH) 2; [OH¯] = 1.0 * 10¯17 M
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The end point in a titration is defined as the point when the indicator changes color.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The solubility of silver chromate is 0.0287 g/1.0 L of solution. What is the Ksp for Ag2CrO4?
A) 9.5 * 10¯5
B) 2.4 * 10¯5
C) 2.6 * 10¯12
D) 6.5 * 10¯13
E) < 1.0 * 10¯13
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is the best representation of the titration curve which will be obtained in the titration of a weak base (0.10 mol L¯1) with HCl of the same concentration?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A solution is prepared by adding 500 mL of 0.3 M NaClO to 500 mL of 0.4 M HClO. What is the pH of this solution?
A) The pH will be greater than the pKa of hypochlorous acid.
B) The pH will be less than the pKa of hypochlorous acid.
C) The pH will be equal to the pKa of hypochlorous acid.
D) The pH will equal the pKb of sodium hypochlorite.
E) None of these choices is correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Citric acid has an acid dissociation constant of 8.4 *10¯4. It would be most effective for preparation of a buffer with a pH of:
A) 2
B) 3
C) 4
D) 5
E) 6
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 25.0-mL sample of 0.35 M HCOOH is titrated with 0.20 M KOH. What is the pH of the solution after 25.0 mL of KOH has been added to the acid? Ka = 1.77 * 10¯4
A) 4.00
B) 3.88
C) 3.63
D) 3.51
E) 3.47
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the following information to calculate the solubility product constant, Ksp, for CuCl. A saturated solution of CuCl in water was prepared and filtered. From the filtrate, 1.0 L was measured out into a beaker and evaporated to dryness. The solid CuCl residue recovered in the beaker was found to weigh 0.041g.
A) Ksp =1.7 *10¯9
B) Ksp = 1.7 *10¯7
C) Ksp = 1.7 *10¯5
D) Ksp = 4.3 *10¯4
E) Ksp = 2.1 *10¯2
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Increasing the concentrations of the components of a buffer solution will increase the buffer capacity.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the solubility of zinc hydroxide, Zn(OH) 2, in 1.00 M NaOH. 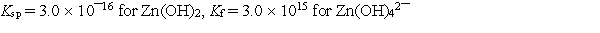
A) 0.60 M
B) 0.52 M
C) 0.37 M
D) 0.32 M
E) 0.24 M
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 50.0-mL sample of 0.50 M HCl is titrated with 0.50 M NaOH. What is the pH of the solution after 28.0 mL of NaOH have been added to the acid?
A) 0.85
B) 0.75
C) 0.66
D) 0.49
E) 3.8
G) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 115
Related Exams