A) [bicarbonate]/[carbonic acid] = 0.11
B) [bicarbonate]/[carbonic acid] = 0.38
C) [bicarbonate]/[carbonic acid] = 2.65
D) [bicarbonate]/[carbonic acid] = 9.4
E) None of these choices are correct.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following substances has the greatest solubility in water?
A) Ba(IO 3) 2, K sp = 1.5 × 10 −9
B) PbF 2, K sp = 3.6 × 10 −8
C) SrSO 4, K sp = 3.2 × 10 −7
D) CuCl, K sp = 1.9 × 10 −7
E) CdS, K sp = 1.0 × 10 −24
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A solution is prepared by dissolving 20.0 g of K2HPO4 and 25.0 g of KH2PO4 in enough water to produce 1.0 L of solution. What is the pH of this buffer? For phosphoric acid (H3PO4) , Ka2 = 6.2 × 10−8.
A) 7.70
B) 7.42
C) 7.21
D) 7.00
E) 6.72
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pKa for the acid HA if a solution of 0.65 M HA and 0.85 M NaA has a pH of 4.75?
A) < 4.00
B) 4.63
C) 4.87
D) 5.02
E) > 5.50
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 25.0-mL sample of 0.35 M HCOOH is titrated with 0.20 M KOH. What is the pH of the solution after 25.0 mL of KOH has been added to the acid? Ka = 1.77 × 10−4
A) 4.00
B) 3.88
C) 3.63
D) 3.51
E) 3.47
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following aqueous solutions, when mixed with an equal volume of 0.10 mol L−1 aqueous NH3, will produce a buffer solution?
A) 0.10 mol L −1 HCl
B) 0.20 mol L −1 HCl
C) 0.10 mol L −1 CH 3COOH
D) 0.050 mol L −1 NaOH
E) 0.20 mol L −1 NH 4Cl
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A buffer is prepared by adding 0.5 mol of solid sodium hydroxide to 1.0 L of 1.0 M acetic acid (CH3COOH) . What is the pH of the buffer?
A) The pH will be pK a - 0.30, where pK a is that of acetic acid.
B) The pH will be greater than the pK a for acetic acid.
C) The pH will be less than the value in answer a.
D) The pH will be equal to the pK a for acetic acid.
E) More information is needed to solve the problem.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the solubility of barium carbonate, BaCO3, in pure water. Ksp = 2.0 × 10−9
A) 1.3 × 10 −3 M
B) 3.2 × 10 −5 M
C) 2.2 × 10 −5 M
D) 4.5 × 10 −5 M
E) 4.0 × 10 −18 M
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the [H3O+] in a solution that consists of 1.5 M NH3 and 2.5 NH4Cl? Kb = 1.8 × 10−5
A) 1.1 × 10 −5 M
B) 3.0 × 10 −6 M
C) 3.3 × 10 −9 M
D) 9.3 × 10 −10 M
E) None of these choices are correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following indicators would be the best to use when 0.050 M benzoic acid (Ka = 6.6 × 10−5) is titrated with 0.05 MNaOH?
A) Bromphenol blue, pH range: 3.0-4.5
B) Bromcresol green, pH range: 3.8-5.4
C) Alizarin, pH range: 5.7-7.2
D) Phenol red, pH range: 6.9-8.2
E) Phenolphthalein, pH range: 8.0-10.1
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is the best representation of the titration curve that will be obtained in the titration of a weak base (0.10 mol L−1) with HCl of the same concentration? 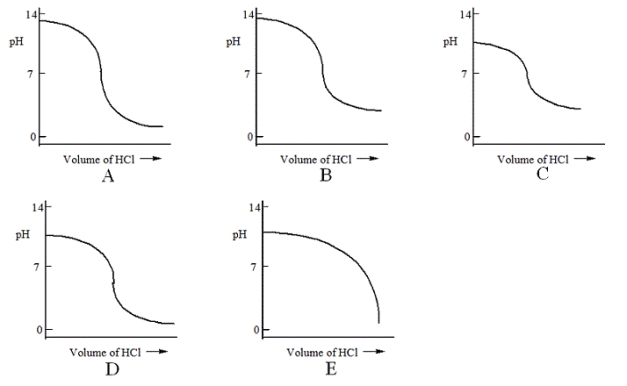
A) A
B) B
C) C
D) D
E) E
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When 0.300 g of a diprotic acid was titrated with 0.100 M LiOH, 40.0 mL of the LiOH solution was needed to reach the second equivalence point. Identify the formula of the diprotic acid.
A) H 2S
B) H 2C 2O 4
C) H 2C 4H 4O 6
D) H 2Se
E) H 2Te
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A diprotic acid H2A has Ka1 = 1 × 10−4 and Ka2 = 1 × 10−8. The corresponding base A2− is titrated with aqueous HCl, both solutions being 0.1 mol L−1. Which one of the following diagrams best represents the titration curve which will be seen? 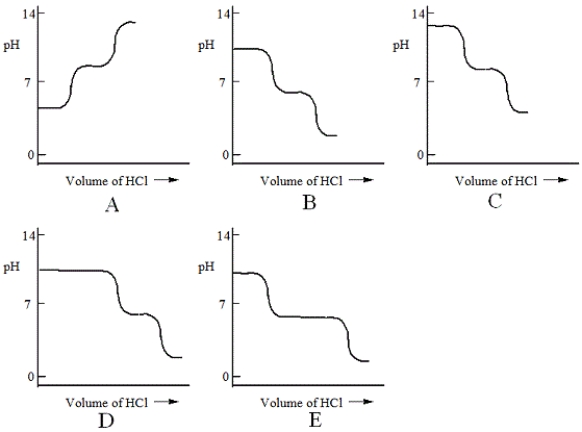
A) A
B) B
C) C
D) D
E) E
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Barium sulfate (BaSO4) is a slightly soluble salt, with Ksp = 1.1 × 10−10. What mass of Ba2+ ions will be present in 1.0 L of a saturated solution of barium sulfate?
A) < 10 −7 g
B) 1.0 × 10 −5 g
C) 0.0014 g
D) 0.0024 g
E) > 0.05 g
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the molar solubility of silver carbonate in 1.0 M sodium carbonate solution. (Ksp for Ag2CO3 = 8.1 x 10-12)
A) 8.1 x 10 -12 M
B) 2.8 x 10 -6 M
C) 1.4 x 10 -6 M
D) 1.4 x 10 -8 M
E) 2.0 x 10 -4 M
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The solubility of magnesium phosphate is 2.27 × 10−3 g/1.0 L of solution. What is the Ksp for Mg3(PO4) 2?
A) 6.5 × 10 −12
B) 6.0 × 10 −14
C) 5.2 × 10 −24
D) 4.8 × 10 −26
E) 1.0 × 10 −26
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the following information to calculate the solubility product constant, Ksp, for PbCl2. A saturated solution of PbCl2 in water was prepared and filtered. From the filtrate, 1.0 L was measured out into a beaker and evaporated to dryness. The solid PbCl2residue recovered in the beaker amounted to 0.0162 moles.
A) K sp = 6.9 × 10 −8
B) K sp = 4.3 × 10 −6
C) K sp = 1.7 × 10 −5
D) K sp = 2.6 × 10 −4
E) K sp = 3.2 × 10 −2
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The solubility of calcium chromate is 1.56 × 10−3 g/100 mL of solution. What is the Ksp for CaCrO4?
A) 2.4 × 10 −4
B) 1.5 × 10 −5
C) 7.6 × 10 −6
D) 1.0 × 10 −8
E) < 1.0 × 10 −8
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following substances has the greatest solubility in water?
A) MgCO 3, K sp = 3.5 × 10 −8
B) NiCO 3, K sp = 1.3 × 10 −7
C) AgIO 3, K sp = 3.1 × 10 −8
D) CuBr, K sp = 5.0 × 10 −9
E) AgCN, K sp = 2.2 × 10 −16
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What will be the effect of adding 0.5 mL of 0.1 M NaOH to 100 mL of an acetate buffer in which [CH3COOH] = [CH3COO−] = 0.5 M?
A) The pH will increase slightly.
B) The pH will increase significantly.
C) The pH will decrease slightly.
D) The pH will decrease significantly.
E) Since it is a buffer solution, the pH will not be affected.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 114
Related Exams