A) -71.4 kJ
B) -286 kJ
C) -357 kJ
D) -428 kJ
E) None of these choices is correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is E°cell for the following reaction? 2Au(s) + 3Ca2+(aq) → 2Au3+(aq) + 3Ca(s) 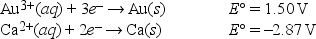
A) -4.37 V
B) -1.37 V
C) -11.6 V
D) 1.37 V
E) 4.37 V
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The voltaic cell composed of Co(s) , Co2+(aq) , Cu(s) , and Cu2+(aq) has E°cell = 0.62 V. If E° of the cathode half-cell is 0.34 V, what is E° of the anode half-cell? Cu2+(aq) + Co(s) → Cu(s) + Co2+(aq)
A) -0.28 V
B) -0.96 V
C) 0.28 V
D) 0.96 V
E) 0.21 V
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the half-reaction at the cathode in a lead storage battery?
A) Pb(s) + PbO2(s) + 4H+(aq) + 2SO42-(aq) → 2PbSO4(s) + 2H2O(l)
B) PbO2(s) + 4H+(aq) + 2SO42-(aq) + 2e- → PbSO4(s) + 2H2O(l)
C) Pb(s) + SO42-(aq) → PbSO4(s) + 2e-
D) Pb(s) → Pb(s) + 2e-
E) H2(g) → 2H+(aq) + 2e-
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many minutes would be required to electroplate 25.0 g of chromium by passing a constant current of 4.80 A through a solution containing CrCl3?
A) 483 min
B) 161 min
C) 322 min
D) 2.01 × 104 min
E) 1.11 × 104 min
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What quantity of charge is required to cause reduction of 0.20 mole of Cr3+ to Cr?
A) 0.60 C
B) 3.0 C
C) 1.9 × 104 C
D) 5.8 × 104 C
E) 9.7 × 104 C
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which equation is correct?
A) Ecell = RT lnK
B) ΔG = -nFEcell
C) Ecell = -RT lnK
D) K = E°cell/nRT
E) E°cell = -nFlnK
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If ΔG° of the following reaction is -110 kJ/mol, what is E°cell? (F = 96,500 C • mol -1) A3+(aq) + 3B(s) → A(s) + 3B+(aq)
A) +0.38 V
B) -0.09 V
C) -0.38 V
D) +0.00038 V
E) +0.09 V
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement is correct?
A) The cathode is the electrode where the oxidation takes place.
B) The cathode is the electrode where the reduction takes place.
C) Both oxidation and reduction make take place at the cathode, depending on the cell.
D) The cathode is always positive.
E) The anode is always negative.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Electrons flow to the cathode in a voltaic cell.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name given to the apparatus where oxidation occurs in a cell where electricity flows?
A) Cathode
B) Electrode
C) Galvanic cell
D) Anode
E) Voltaic cell
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Based on the data presented below, which is the strongest reducing agent? 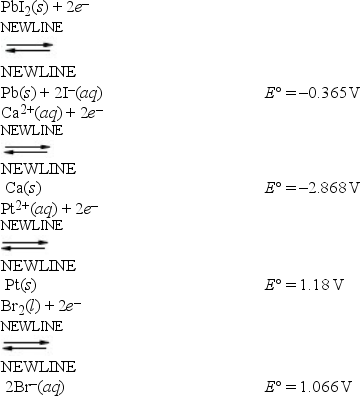
A) Pb(s)
B) Ca(s)
C) Pt(s)
D) Br-(aq)
E) Pt2+(aq)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
________ is the process that uses electrical energy to drive a nonspontaneous chemical reaction.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A voltaic cell consists of a Cd/Cd2+ electrode (E° = -0.40 V) and a Fe/Fe2+ electrode (E° = -0.44 V) . If Ecell = 0 and the temperature is 25°C, what is the ratio [Fe2+]/[Cd2+]?
A) 2.3 × 101
B) 1.0 × 101
C) 1.0
D) 1.0 × 10-1
E) 5.0 × 10-2
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which metals may be oxidized by H+ under standard-state conditions? 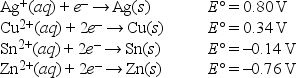
A) Ag, Cu, Sn, and Zn
B) Ag and Cu
C) Sn and Cu
D) Zn and Cu
E) Zn and Sn
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What mass of oxygen gas is produced in the electrolysis of water, for every gram of hydrogen gas formed? Reaction: 2H2O(l) → 2H2(g) + O2(g)
A) 32 g O2
B) 16 g O2
C) 64 g O2
D) 20 g O2
E) 8 g O2
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the correct cell diagram for the following electrochemical cell? 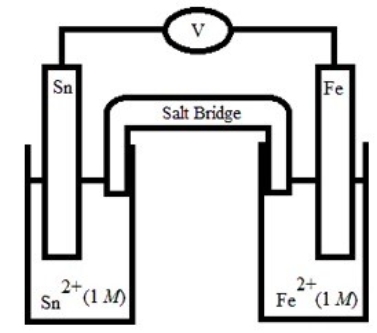
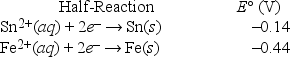
A) Sn(s) | Sn2+(aq, 1.0 M) || Fe2+(aq, 1.0 M) | Fe(s)
B) Sn(s) | Fe2+(aq, 1.0 M) || Sn2+(aq, 1.0 M) | Fe(s)
C) Fe(s) | Sn2+(aq, 1.0 M) || Fe2+(aq, 1.0 M) | Sn(s)
D) Fe(s) | Fe2+(aq, 1.0 M) || Sn2+(aq, 1.0 M) | Sn(s)
E) Sn(s) | Fe(s) || Sn2+(aq, 1.0 M) | Fe2+(aq, 1.0 M)
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following balanced redox reaction. Mn2+(aq) + S2O82-(aq) + 2H2O(l) → MnO2(s) + 4H+(aq) + 2SO42-(aq) Which of the following statements is true?
A) Mn2+(aq) is the oxidizing agent and is reduced.
B) Mn2+(aq) is the oxidizing agent and is oxidized.
C) Mn2+(aq) is the reducing agent and is oxidized.
D) Mn2+(aq) is the reducing agent and is reduced.
E) Manganese does not change its oxidation number in this reaction.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is ΔG° at 200°C for the following reaction? (F = 96,500 C • mol -1) 2Na(l) + FeCl2(s)  2NaCl(s) + Fe(s) E°cell = 2.35 V
2NaCl(s) + Fe(s) E°cell = 2.35 V
A) 454 kJ/mol
B) -454 kJ/mol
C) 907 kJ/mol
D) -907 kJ/mol
E) 227 kJ/mol
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is E°cell for a galvanic cell represented by the combination of the following half-reactions? 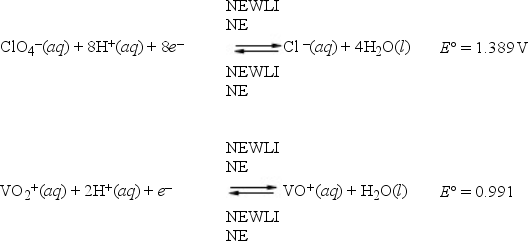
A) -0.398 V
B) -2.380 V
C) 0.398 V
D) 2.380 V
E) 6.539 V
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 101 - 120 of 122
Related Exams