Correct Answer

verified
A)11ec6d40_aba9_d0ba_9cd1_27df90ce31a5_...View Answer
Show Answer
Correct Answer
verified
A)11ec6d40_aba9_d0ba_9cd1_27df90ce31a5_...
View Answer
Multiple Choice
Predict the actual bond angles in SF3+ using the VSEPR theory.
A) more than 120°
B) exactly 120°
C) between 109° and 120°
D) between 90° and 109°
E) less than 90°
G) A) and B)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Use VSEPR theory to decide which one of the following ions and molecules is likely to be planar. (The central atom is always first in the formula.)
A) BrF3
B) H3O+
C) PCl3
D) SO42-
E) SF4
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Considering all the bonds in a molecule with trigonal bipyramidal geometry, what are the bond angles present?
A) 120° only
B) 90° only
C) 180° only
D) 60° and 90° only
E) 90°, 120°, and 180°
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following Lewis structures is definitely incorrect?
A) NO ![]()
B) HCN ![]()
C) NO2- ![]()
D) SO32- ![]()
E) PCl5 ![]()
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which one of the following species is the central atom (the first atom in the formula) likely to violate the octet rule?
A) BF4-
B) XeO3
C) SiCl4
D) NH3
E) CH2Cl2
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory, a molecule with the general formula AX2 will have a ___ molecular shape.
A) linear
B) bent
C) trigonal planar
D) tetrahedral
E) triangular
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the following Lewis structure for phosphate, phosphorus has a formal charge of ____ and an oxidation number of ____. 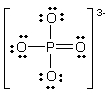
A) 0, -3
B) 0, 5
C) 5, -3
D) 5, 5
E) 3, 5
G) All of the above
Correct Answer

verified
Correct Answer
verified
Essay
What is the shape of the PF3 molecule? Explain your answer, using VSEPR theory.
Correct Answer

verified
The Lewis structure has a lone pair on t...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which of the following atoms can expand its valence shell when bonding?
A) N
B) C
C) O
D) P
E) Al
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of ClF4- as predicted by the VSEPR theory?
A) square pyramidal
B) square planar
C) see-saw
D) octahedral
E) tetrahedral
G) A) and E)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
In which one of the following is the best Lewis structure a resonance structure?
A) SO3
B) BF3
C) I3-
D) SCO (C = central atom)
E) SO32-
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
A molecule which contains polar bonds will always have a dipole moment.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use VSEPR theory to decide which one of the following molecules and ions will have a trigonal pyramidal geometry. (The central atom is always first in the formula.)
A) PCl3
B) BF3
C) SO3
D) BrF3
E) CO32-
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of SCl3F as predicted by the VSEPR theory?
A) linear
B) bent
C) see-saw
D) T-shaped
E) trigonal pyramidal
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following has no net dipole moment?
A) N2O
B) NF3
C) H2Se
D) TeO3
E) CH3Cl
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the COCl2 molecule, carbon is the central atom. Based on the best Lewis structure for COCl2, what is the formal charge on carbon?
A) 0
B) +1
C) -1
D) +2
E) -2
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
List all possible molecular geometries (shapes) for a nonpolar molecule with the formula AX4.
A) tetrahedral
B) seesaw
C) square planar
D) either tetrahedral or square planar
E) any of the above shapes
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In a Lewis structure for a molecule or ion, the sum of the formal charges on the atoms is equal to the charge on the molecule or ion.
B) False
Correct Answer

verified
Correct Answer
verified
Essay
Draw all important resonance structures of the nitrate ion, NO3-
Correct Answer

verified
11ea8ef8_2b58_6b95_ab0a_fb70444a8199_TB5833_00
Correct Answer
verified
Showing 1 - 20 of 109
Related Exams