A) CH3OCH2CH2OCH3
B) CH3OCH2OCH2CH3
C) HOCH2CH2CH2CH2OH
D) CH3OCH2CH2CH2OH
E) (CH3O) 2CHCH3
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The compound aspartame is a dipeptide that is often used as a sugar substitute.Which functional groups are present in aspartame? 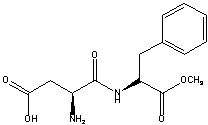
A) carboxylic acid,secondary amine,ketone,ester
B) alcohol,secondary amine,ketone,ester
C) carboxylic acid,ester,amide,secondary amine
D) ester,amide,primary amine,carboxylic acid
E) none of these choices
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The strongest of attractive forces is which type?
A) Dispersion forces
B) Ion-dipole
C) Dipole-dipole
D) Cation-anion
E) Hydrogen bonds
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which molecule has a dipole moment of zero?
A) CHCl3
B) CH2Cl2
C) ClHC=CH2
D) trans-ClHC=CHCl
E) None of these choices.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What alkyl groups make up the following ketone? 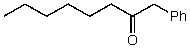
A) Phenyl,pentyl
B) Hexyl,phenyl
C) Benzyl,hexyl
D) Benzyl,heptyl
E) None of these choices.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which compound would you expect to have the highest boiling point?
A) ethyl alcohol
B) ethyl amine
C) chloroethane
D) water
E) ethane
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a molecule to possess a dipole moment,which following condition is necessary but not sufficient?
A) three or more atoms in the molecule
B) presence of one or more polar bonds
C) a non-linear structure
D) presence of oxygen or fluorine
E) absence of a carbon-carbon double or triple bond
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What alkyl groups make up the following ether? 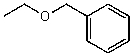
A) ethyl and phenyl
B) propyl and benzyl
C) ethyl and benzyl
D) propyl and phenyl
E) None of these choices.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What functional groups are present in nicotine,an addictive substance found in tobacco? 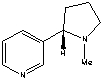
A) two tertiary amines
B) two secondary amines
C) phenyl ring and tertiary amine
D) secondary and tertiary anime
E) none of these choices
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which molecule has a zero dipole moment?
A) SO2
B) CO2
C) CO
D) CHCl3
E) None of these choices.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What group makes up the following aldehyde (benzaldehyde) ? 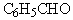
A) benzyl
B) phenyl
C) heptyl
D) ethyl
E) None of these choices.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An oxygen-containing compound shows strong IR absorption at 1630-1780 cm-1 and 3200-3550 cm-1.What type of compound is it likely to be?
A) an alcohol
B) a carboxylic acid
C) an ether
D) a ketone
E) an aldehyde
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the functional group(s) on the following molecule,what characteristic IR absorption(s) would be expected (ignoring C-H absorptions) ? 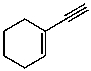
A) peaks around 3300,2150,and 1650 cm-1
B) peaks around 1710 and 1650 cm-1
C) peaks around 1650 and 3300 cm-1
D) a peak around 2250 and 3300 cm-1
E) None of these choices.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Draw a structural formula for C8H18,in which there are two quaternary carbons.
Correct Answer

verified
Correct Answer
verified
Short Answer
Examine the following IR spectrum,for substance P (C3H5NO).Which oxygen containing functional group is most likely present in P? 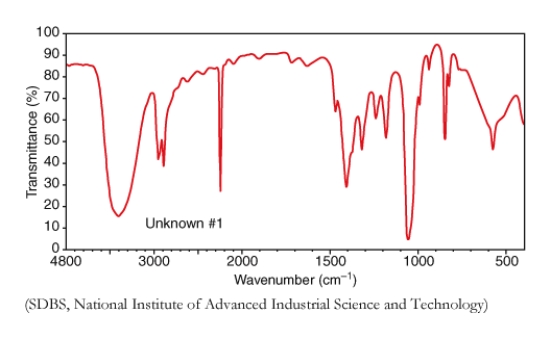
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following reaction sequence (it is not necessary to understand the chemistry) what significant overall change(s) would be expected by IR (ignoring C-H absorptions) ? 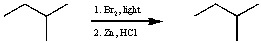
A) A peak around 3300 cm-1 would disappear and nothing new would appear.
B) A peak around 1710 cm-1 would disappear and a new peak around 3300 cm-1 would appear.
C) A peak around 1650 cm-1 would disappear and nothing new would appear.
D) No overall change would be observed.
E) None of these choices.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which molecule does not have a dipole moment?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) None of these choices.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Many bacterial cells contain functional groups that are susceptible to antibiotic drugs,but may,over time,change their functional groups to become resistant to these drugs.What functional group change is occurring in the transition shown below? 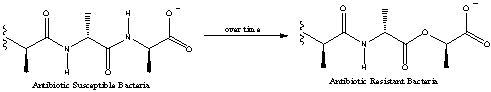
A) Amine to ether
B) Amine to ester
C) Amide to ester
D) Amide to ether
E) None of these choices.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What functional group(s) is/are present in the following compound? 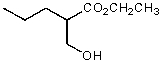
A) Ketone and 1o alcohol
B) Ether and alcohol
C) Ester and ether
D) Ester and 1o alcohol
E) 1o alcohol and aldehyde
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An example of a tertiary amine is: 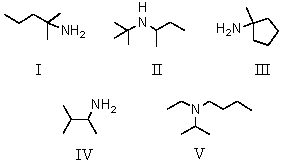
A) I
B) II
C) III
D) IV
E) V
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Showing 101 - 120 of 151
Related Exams